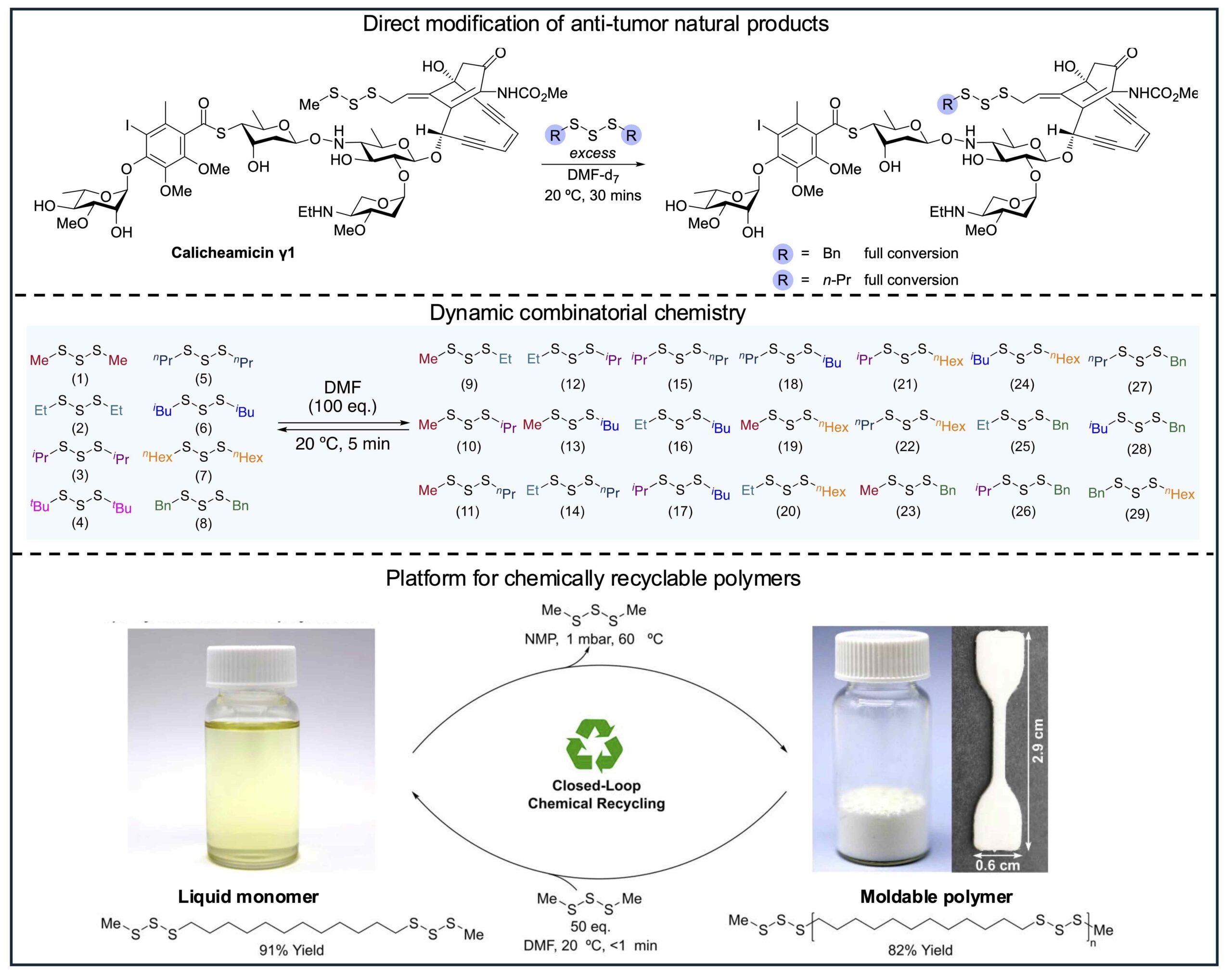
International researchers have confirmed the discovery of a groundbreaking chemical reaction that holds promise for various sectors, including recycled plastics and pharmaceuticals. This new reaction, known as the trisulfide metathesis reaction, enables the spontaneous formation and breaking of sulfur-sulfur (S-S) bonds without requiring external reagents or stimuli. The implications of this discovery could lead to rapid advancements in numerous applications.
The research, led by Professor Justin Chalker from Flinders University, began over a decade ago with a focus on developing environmentally friendly sulfur polymers. Professor Chalker noted, “It is rare to discover an entirely new reaction, and even more rare for it to be useful in so many fields and applications.” This new reaction has already demonstrated its effectiveness in modifying an anti-tumor drug and creating a novel plastic that can be easily recycled.
The S-S bonds, which are crucial components in peptides, proteins, and various polymers like vulcanised rubber, have traditionally been challenging to manipulate. The trisulfide metathesis reaction stands out due to its high reaction rates and selectivity, allowing for a cleaner and more efficient process. In many cases, the reaction completes within seconds, significantly enhancing the potential for industrial applications.
In particular, the ability to modify compounds such as calicheamicin, an anti-cancer drug containing a trisulfide, marks a significant step forward in the development of targeted therapies. According to Dr Harshal Patel, the first author of the study, this novel reaction has been successfully applied to modify anti-cancer drugs and create a chemical library relevant to drug discovery. “I’m excited to see how this chemistry is adopted, expanded and applied in ways not yet imagined,” Dr Patel stated.
The research team, which also includes Dr Tom Hasell from the University of Liverpool, began their explorations with funding from the Australian Research Council (ARC). Their initial findings revealed unexpected behaviors of S-S bonds in certain solvents. Further investigations at Flinders University, involving multiple researchers from both Australia and the UK, led to the development of a mechanistic model explaining how these bonds break and reform.
This foundational understanding paves the way for various applications, including the selective modification of natural products, rapid synthesis of compound libraries relevant to medicinal chemistry, and the creation of fully recyclable polymers. The potential to produce recyclable versions of polyethylene is particularly noteworthy, as it aligns with global sustainability efforts. A subsequent ARC grant will support further exploration into the applications of this chemistry for recyclable plastics, rubber, foam, and fibers.
Dr Hasell emphasized the versatility of the trisulfide metathesis reaction, stating, “I think the examples we’ve shown of what can be done with this chemistry are only the tip of the iceberg.” The research team, including PhD researcher James Smith and postdoctoral associate Dr Patel, is set to continue exploring this innovative chemistry.
The study, titled “Spontaneous Trisulfide Metathesis in Polar Aprotic Solvents,” will be published in Nature Chemistry in March 2026. The research received significant financial support from the Australian Research Council, underscoring the importance of this discovery for future advancements in chemical science and sustainability.