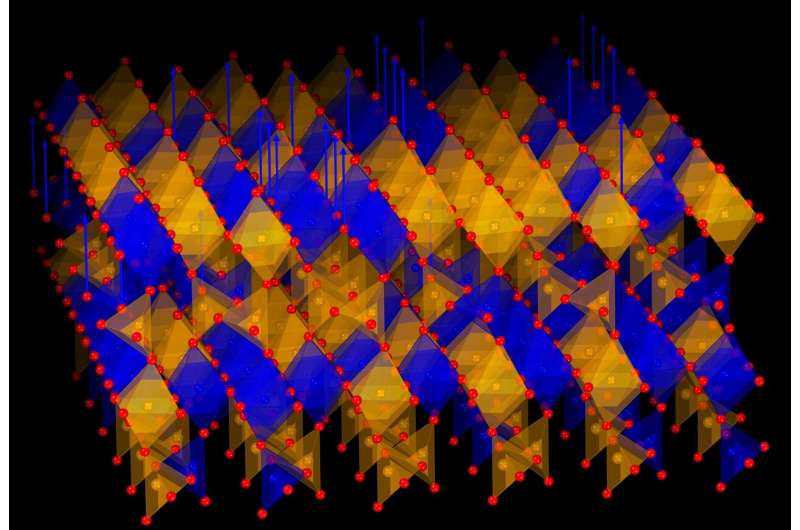
A groundbreaking discovery has emerged from a collaborative research effort between scientists in Korea and Japan. A team has developed a new type of crystal that exhibits the remarkable ability to “breathe” oxygen, absorbing and releasing it repeatedly at relatively low temperatures. This innovative material has the potential to significantly advance clean energy technologies, including fuel cells, energy-efficient windows, and smart thermal devices.
The newly discovered crystal comprises a special metal oxide made of strontium, iron, and cobalt. Its unique characteristic allows it to release oxygen when heated in a simple gas environment, subsequently absorbing it again without structural degradation. This process can occur multiple times, making the material suitable for practical applications across various industries.
Significance of the Discovery
Led by Professor Hyoungjeen Jeen from the Department of Physics at Pusan National University in Korea, and co-authored by Professor Hiromichi Ohta from the Research Institute for Electronic Science at Hokkaido University in Japan, the study highlights a significant advancement in material science. Their findings, published in the journal Nature Communications on August 15, 2025, emphasize the ability of this crystal to mimic respiratory functions.
“It is like giving the crystal lungs; it can inhale and exhale oxygen on command,” said Professor Jeen. This capacity to control oxygen is pivotal for technologies such as solid oxide fuel cells, which generate electricity from hydrogen with minimal emissions, and thermal transistors that manage heat flow like electrical switches.
Until now, most materials capable of oxygen control were either too fragile or only functional under extreme conditions. The new crystal operates effectively under milder temperatures, maintaining its stability throughout the process.
Reversible Process and Future Applications
The research team demonstrated that the material could revert to its original structure when oxygen is reintroduced, confirming the reversibility of the process. “This is a major step toward the realization of smart materials that can adjust themselves in real time,” Professor Ohta noted.
The implications of this research extend beyond clean energy. The potential applications encompass electronics and eco-friendly building materials, indicating a broad scope for future innovations. As the world increasingly seeks sustainable solutions, such advancements could play a crucial role in reducing environmental impacts.
In summary, the discovery of this new crystal represents a significant leap forward in material science, with the ability to influence various sectors from energy to construction. The ongoing exploration of its capabilities will likely unveil further opportunities for sustainable development in the coming years.
For more detailed insights, refer to the original study: Joonhyuk Lee et al, “Selective reduction in epitaxial SrFe 0.5 Co 0.5 O 2.5 and its reversibility,” published in Nature Communications. DOI: 10.1038/s41467-025-62612-1.