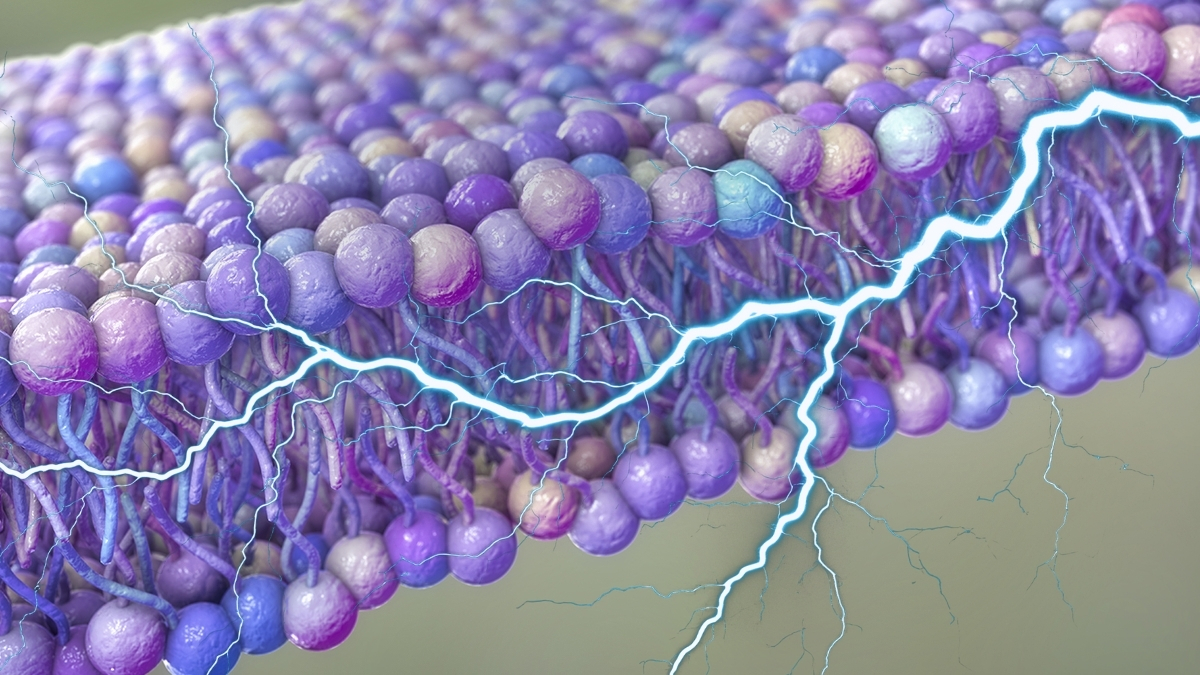
Researchers from the University of Houston and Rutgers University have identified a previously unrecognized source of electrical power within cells. Their study suggests that small ripples in the fatty membranes surrounding cells may generate sufficient voltage to facilitate critical biological processes, possibly enhancing cellular communication and material transport.
The investigation builds on prior research concerning the activity of embedded proteins and the breakdown of adenosine triphosphate (ATP), the primary energy carrier in cells. The researchers propose that these membrane fluctuations could create an electric charge strong enough to be harnessed for essential cellular functions.
Understanding Flexoelectricity and Its Implications
Central to this new model is the concept of flexoelectricity, which refers to the generation of voltage through varying points of strain within a material. The membranes of cells are not static; they constantly bend due to random heat fluctuations. In theory, any voltage produced from these movements would negate itself in a state of equilibrium. However, the researchers argue that the dynamic nature of cellular activity prevents strict equilibrium, thus allowing these fluctuations to produce usable energy.
The team calculated that flexoelectricity could create a voltage difference across the cell membrane of up to 90 millivolts. This voltage level is sufficient to trigger a neuron’s firing, indicating that the fluctuations could significantly influence ion transport—the movement of charged atoms essential for various cellular activities. The researchers estimate that these charges emerge on a millisecond scale, aligning perfectly with the timing of signals transmitted through nerve cells.
The findings suggest that membrane fluctuations could facilitate vital biological operations, including muscle movement and sensory processing. The research team notes, “Our results reveal that activity can significantly amplify transmembrane voltage and polarization, suggesting a physical mechanism for energy harvesting and directed ion transport in living cells.”
Broader Implications and Future Research
This discovery could have far-reaching implications, not just for understanding cellular function but also for potential applications beyond biological systems. The researchers propose that the principles of electricity generation identified in living cells could inform the development of artificial intelligence networks and synthetic materials inspired by natural designs.
They state, “Investigating electromechanical dynamics in neuron networks may bridge molecular flexoelectricity and complex information processing, with implications for both understanding brain function and discovering bio-inspired computational materials.”
The research has been published in the journal PNAS Nexus, opening avenues for future studies to explore these phenomena within living organisms. Further investigations could validate the theoretical underpinnings of their findings and explore how this newly discovered power source can be harnessed in both biological and synthetic contexts.