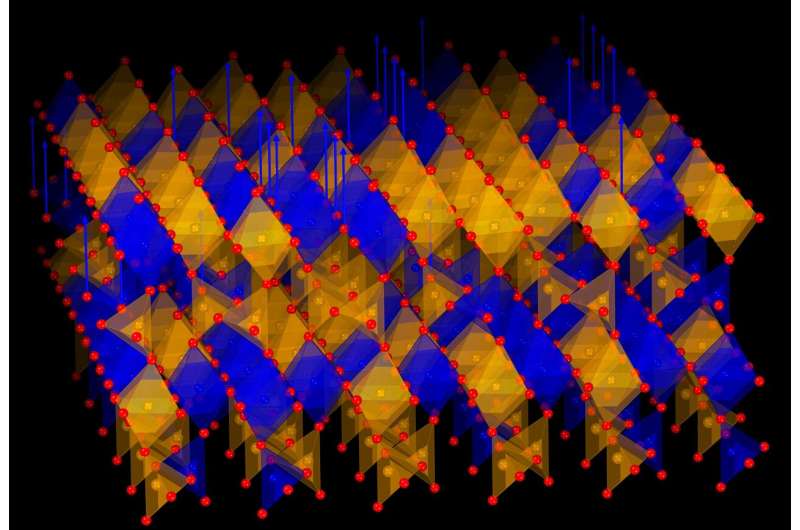
A collaborative team of scientists from Korea and Japan has made a significant breakthrough with the discovery of a new type of crystal that can effectively “breathe” oxygen. This innovative material can absorb and release oxygen repeatedly at relatively low temperatures, which could pave the way for advancements in clean energy technologies such as fuel cells, energy-efficient windows, and smart thermal devices. The findings were published in the journal Nature Communications on August 15, 2025.
The newly developed material is a specific kind of metal oxide composed of strontium, iron, and cobalt. Its remarkable characteristic is its ability to release oxygen when heated in a simple gas environment and then reabsorb it without structural degradation. This reversible process can be repeated multiple times, making the crystal suitable for various practical applications.
Leading the research is Professor Hyoungjeen Jeen from the Department of Physics at Pusan National University in Korea, with co-authorship from Professor Hiromichi Ohta of the Research Institute for Electronic Science at Hokkaido University, Japan.
The ability to control oxygen in materials is vital for technologies like solid oxide fuel cells, which generate electricity from hydrogen while minimizing emissions. Additionally, it is applicable in thermal transistors—devices that can manage heat like electrical switches—and smart windows that adapt their thermal properties based on environmental conditions.
Historically, materials that exhibited this level of oxygen control were often too fragile or only functional under extreme conditions, such as very high temperatures. This new crystal, however, operates effectively under milder environments while maintaining stability.
Professor Jeen elaborated on the significance of the discovery, stating, “This finding is striking in two ways: only cobalt ions are reduced, and the process leads to the formation of an entirely new but stable crystal structure.” The researchers demonstrated that the material could revert to its original state upon the reintroduction of oxygen, confirming the process’s full reversibility.
“This is a major step toward the realization of smart materials that can adjust themselves in real time,” noted Professor Ohta. He emphasized the extensive potential applications of this discovery, which range from clean energy solutions to innovations in electronics and eco-friendly construction materials.
As the world seeks sustainable solutions to energy and environmental challenges, the implications of this research could be profound. The ability to harness and manipulate oxygen in such a versatile manner positions this crystal as a promising candidate for future technological advancements.
For further details, refer to the study titled “Selective reduction in epitaxial SrFe0.5Co0.5O2.5 and its reversibility,” published in Nature Communications.